 It is the only non-metal that is a liquid at normal room conditions. It was first isolated in pure form by Balard in 1826. 
In fact its name is derived from the Greek bromos or "stench". Chemistry of Bromine (Z=35) Bromine is a reddish-brown fuming liquid at room temperature with a very disagreeable chlorine-like smell.Due to its high reactivity, it is commonly found in nature bonded to many different elements. It is very reactive and is widely used for many purposes, such as as a disinfectant. Chemistry of Chlorine (Z=17) Chlorine is a halogen in group 17 and period 3.It is a nonmetal, and is one of the few elements that can form diatomic molecules (F2). It is the most electronegative element, given that it is the top element in the Halogen Group, and therefore is very reactive. Its atomic number is 9 and its atomic weight is 19, and it's a gas at room temperature. Chemistry of Fluorine (Z=9) Fluorine (F) is the first element in the Halogen group (group 17) in the periodic table.Oxidizing Ability of the Group 17 Elements.Includes trends in atomic and physical properties, the redox properties of the halogens and their ions, the acidity of the hydrogen halides, and the tests for the halide ions. Group 17: Chemical Properties of the Halogens Covers the halogens in Group 17: fluorine (F), chlorine (Cl), bromine (Br) and iodine (I).Physical Properties of the Group 17 Elements.Group 17: General Properties of Halogens.Atomic and Physical Properties of Halogens.As a result, there is a regular increase in the ability to form high oxidation states. On the other hand, there is a regular decrease in the first ionization energy as we go down this group. This includes their melting points, boiling points, intensity of their color, the radius of the corresponding halide ion, and the density of the element. Group 17: Physical Properties of the Halogens It can be seen that there is a regular increase in many of the properties of the halogens proceeding down group 17 from fluorine to iodine.These examples are programmatically compiled from various online sources to illustrate current usage of the word 'noble gas.' Any opinions expressed in the examples do not represent those of Merriam-Webster or its editors. Jennifer Ouellette, Ars Technica, 2 June 2022 See More Joshua Hawkins, BGR, 7 July 2022 However, work by other research teams in 2015 ruled out a comet or meteorite as the stone's source, based on noble gas and nuclear probe analyses. 2019 Xenon is a colorless and odorless noble gas that is found in tiny amounts throughout our atmosphere. 2022 The other option is to use a noble gas, such as neon. Cody Cottier, Discover Magazine, 12 Apr. 2019 Scerri, for one, is a fan, and notes that helium’s counterintuitive position may yet be vindicated - in 2017, the supposedly noble gas was found to form a stable compound with sodium at high pressure. Ken Croswell, Discover Magazine, 12 Dec. 2014 Another noble gas element helped to make the find possible. 2023 Your chemistry teacher probably taught you that helium, a noble gas, never reacts with anything, but that turns out to be entirely untrue - at least, under certain highly exotic conditions. 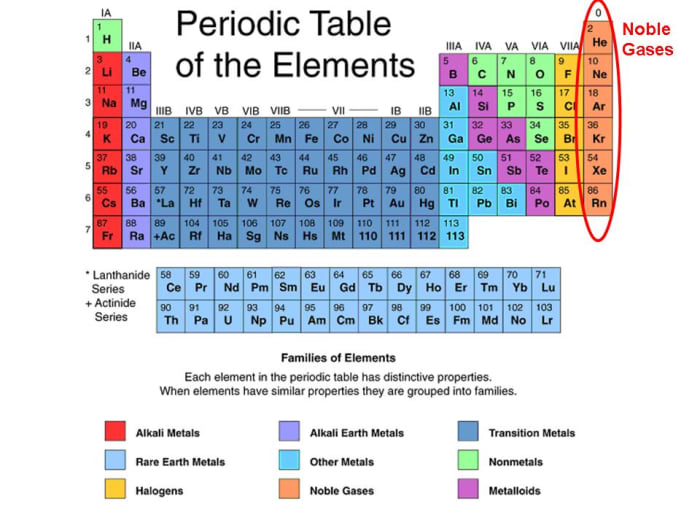
Lina Abascal, Los Angeles Times, 22 Aug. 2023 The noble gas neon was first isolated by British chemists at the turn of the 20th century. Recent Examples on the Web Chemists group elements with similar properties, like halogens and noble gases. 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
